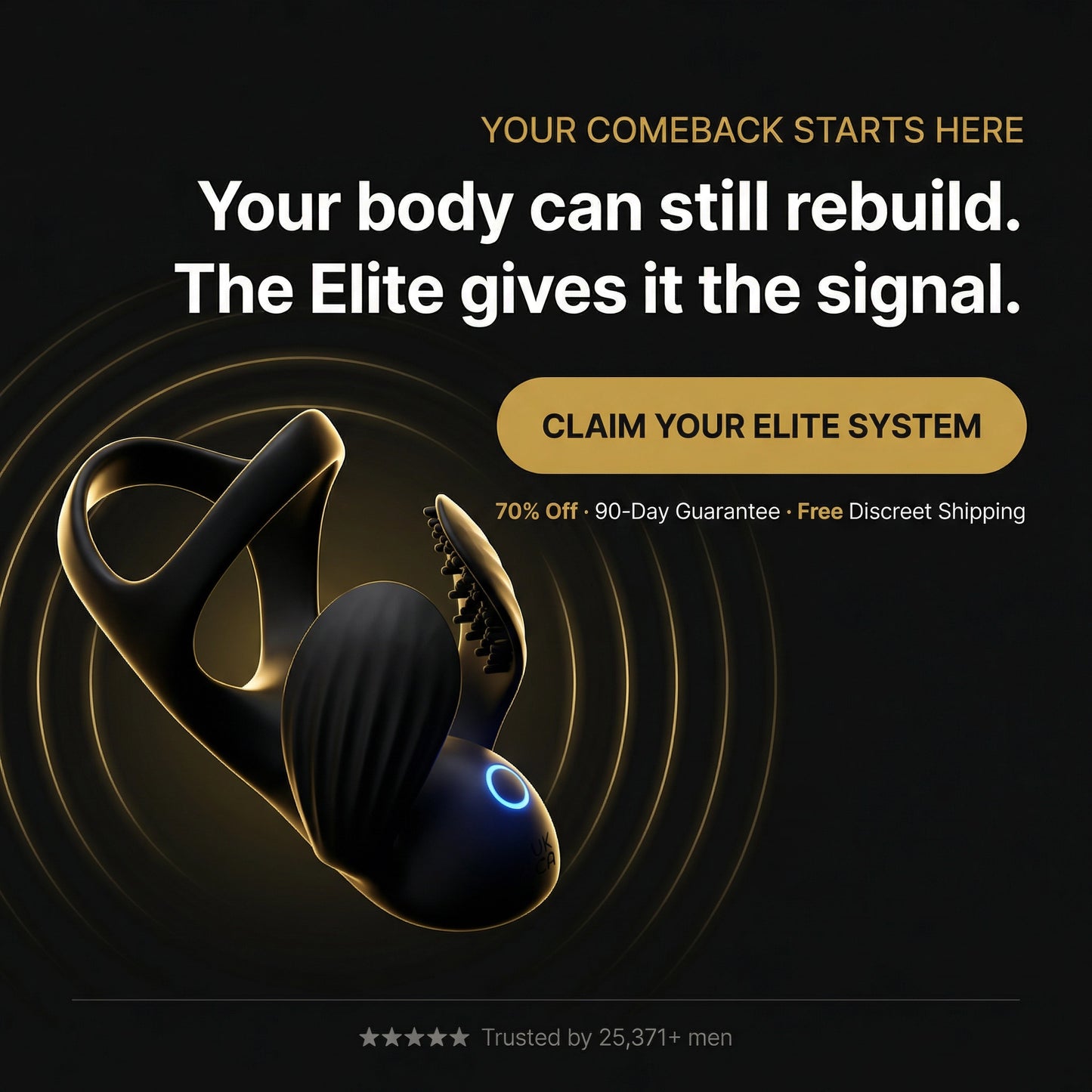
Acoustic Shockwave Therapy
Micro-pulses stimulate new blood vessel growth — restoring natural circulation where it matters most.
Spend $70 more to get FREE shipping!

Replace Your Product Anytime For Life | Limit 1 Use Per Customer

78% Of Users Pair Their Device With The 90 Day Master Lover Program For Faster Confidence, Lasting Stamina, Complete Bedroom Mastery.

Premium Blend of Shilajit, Ashwaghanda, Seamoss | Boost Testosterone Naturally | FDA-Approved























Both devices use the same clinical-grade shockwave technology. The difference is how fast you get there.

Acoustic Wave Therapy

Acoustic Wave + PEMF — 2× Faster Results
Developed by a team of urologists and biomedical engineers. The only dual-therapy device built to support healthy blood flow and nerve response — no pills, no clinic visits.

What's Really Happening Inside Your Body
It's not age. It's biology — and biology can be rebuilt. Here's the progression most men don't see until it's too late.

Blood vessels lose flexibility. Nitric oxide drops. Micro-circulation falls 15–20% — and you start blaming stress for what’s actually vascular decline already in motion.

Acoustic wave therapy was locked behind clinic doors — $500+ per session, 6+ visits. ApexDrive Elite delivers the same dual-therapy protocol at home for a fraction of the cost. Same science, complete privacy.

Micro-pulses stimulate new blood vessel growth — restoring natural circulation where it matters most.
Electromagnetic fields activate cellular repair and nerve regeneration — amplifying sensitivity and recovery.
Shockwave + PEMF combined deliver up to 52% stronger results than single-mode devices. 82% clinical efficacy.
All three therapies converge on one critical molecule — nitric oxide. This vasodilator relaxes smooth muscle, widens blood vessels, and restores the natural erectile response from within.
Low-intensity acoustic pulses trigger angiogenesis — the growth of new blood vessels — restoring natural circulation to the tissue that matters most.
Pulsed electromagnetic fields penetrate deep tissue to reactivate dormant nerve pathways and accelerate cellular repair. Sensitivity improves progressively.
Multi-frequency vibration protocols train neuromuscular control — building lasting endurance and timing through consistent use.
Backed by Science,
Trusted by Experts
"Acoustic wave therapy has been used in urology clinics for over a decade. The ability for men to now access clinic-grade shockwave combined with PEMF therapy at home — that represents a real shift in proactive male wellness."

Follow the protocol. Track the changes. Here's the timeline thousands of men experienced.
*Individual results vary. Full 90-day guarantee covers entire protocol cycle.
Triggers new blood vessel growth + nerve regeneration. Addresses the root cause.
Masks symptoms for 4-6 hours. Does nothing for the underlying cause.
Structural changes sustained 12+ months. One device. One investment.
Effects wear off in hours. $30–70/month, every month, indefinitely.
73% of partners noticed natural improvement within 30 days. No planning required.
Partner notices pill timing, mood swings, and the "planning" around intimacy.
No prescription needed. Ships to your door. Start tonight.
Requires doctor visit, prescription, and ongoing refills.
One-time investment. 90-day guarantee. No recurring fees. Ever.
$30–70/month. $360–840/year. Every year.
Triggers new blood vessel growth + nerve regeneration. Addresses the root cause.
Masks symptoms for 4-6 hours. Does nothing for the underlying cause.
Structural changes sustained 12+ months. One device. One investment.
Effects wear off in hours. $30–70/month, every month, indefinitely.
73% of partners noticed natural improvement within 30 days. No planning required.
Partner notices pill timing, mood swings, and the "planning" around intimacy.
No prescription needed. Ships to your door. Start tonight.
Requires doctor visit, prescription, and ongoing refills.
One-time investment. 90-day guarantee. No recurring fees. Ever.
$30–70/month. $360–840/year. Every year.
"The dual-therapy mechanism — shockwave combined with PEMF — aligns with what we see in advanced clinical protocols. I now mention this to patients asking about non-pharmaceutical options with real physiological action."
"Three weeks in, I woke up with something I hadn't felt in months. By week eight, my wife said I was the man she married. Her words, not mine."
"No pills, no planning, no timing anything. Three times a week, fifteen minutes. My body just works again. That freedom is worth more than what I paid."
"I was spending $400 a session at the clinic for shockwave alone. This does shockwave AND PEMF for the price of one visit. The math sold me. The results — and my wife's reaction — sealed it."
"The dual therapy made a real difference in sensitivity. I actually feel everything now — and I still last longer. That's not a trade-off. That's the whole point."
"Went from maybe 60% firmness on a good day to full strength in five weeks. The PEMF pulse is what makes this different — you actually feel it working. Game changer."
"At 67 I figured my best years were behind me. Two months with the Elite and my wife and I are closer than we've been in a decade. She says I'm more present. That means everything."
"I track everything. Morning firmness improved by week two. Full sessions without assistance by week six. The dual mechanism isn't marketing — it's measurable. I have the data."
"Wasn't going to any clinic. Wasn't having that conversation. Fifteen minutes, few times a week, right here at home. Nobody needs to know. And it works. That's all that matters."
| Device Classification: | FDA-Registered Class II Wellness Device — Acoustic + Electromagnetic Dual-Therapy System |
|---|---|
| Technology Platform: | Tri-Modal Convergence™ — Focused Acoustic Shockwave (Li-ESWT) + Pulsed Electromagnetic Field (PEMF) + Targeted Micro-Vibration |
| Medical-Grade Material: | Platinum-Cured LSR Silicone (ISO 10993 Biocompatible, Hypoallergenic, BPA/Phthalate-Free) |
| Internal Housing: | Aircraft-Grade Aluminum Alloy (6061-T6) — EMI Shielded |
| Processor: | 32-Bit ARM Cortex-M4 Microcontroller with Real-Time Therapy Sequencing |
| Battery: | 2,200mAh Medical-Grade Li-Po — 4+ Hours Continuous Use (≈16 sessions per charge) |
| Charging: | USB-C Magnetic Dock — Full Charge in 90 Minutes |
| Weight: | 128g (4.5 oz) — Ergonomic Dual-Ring Form Factor |
| Inner Diameter Range: | 28mm – 45mm (Adjustable Stretch-Fit, accommodates 90th percentile) |
| Water Resistance: | IPX5 Rated — Splash & Sweat Proof (do not submerge) |
| Connectivity: | Bluetooth 5.2 LE — Companion App (iOS 15+ / Android 12+) |
| Operating Temperature: | 15°C – 40°C (59°F – 104°F) |
| Certifications: | CE Marked · FCC Part 15B · RoHS Compliant · ISO 13485 Manufacturing |
| Country of Engineering: | Designed in USA — Precision Manufactured in ISO 13485 Certified Facility |
| Warranty: | 2-Year Manufacturer Warranty + 90-Day Risk-Free Trial |
Device Classification:
FDA-Registered Class II Wellness Device — Acoustic + Electromagnetic Dual-Therapy System
Technology Platform:
Tri-Modal Convergence™ — Focused Acoustic Shockwave (Li-ESWT) + Pulsed Electromagnetic Field (PEMF) + Targeted Micro-Vibration
Medical-Grade Material:
Platinum-Cured LSR Silicone (ISO 10993 Biocompatible, Hypoallergenic, BPA/Phthalate-Free)
Internal Housing:
Aircraft-Grade Aluminum Alloy (6061-T6) — EMI Shielded
Processor:
32-Bit ARM Cortex-M4 Microcontroller with Real-Time Therapy Sequencing
Battery:
2,200mAh Medical-Grade Li-Po — 4+ Hours Continuous Use (≈16 sessions per charge)
Charging:
USB-C Magnetic Dock — Full Charge in 90 Minutes
Weight:
128g (4.5 oz) — Ergonomic Dual-Ring Form Factor
Inner Diameter Range:
28mm – 45mm (Adjustable Stretch-Fit, accommodates 90th percentile)
Water Resistance:
IPX5 Rated — Splash & Sweat Proof (do not submerge)
Connectivity:
Bluetooth 5.2 LE — Companion App (iOS 15+ / Android 12+)
Operating Temperature:
15°C – 40°C (59°F – 104°F)
Certifications:
CE Marked · FCC Part 15B · RoHS Compliant · ISO 13485 Manufacturing
Country of Engineering:
Designed in USA — Precision Manufactured in ISO 13485 Certified Facility
Warranty:
2-Year Manufacturer Warranty + 90-Day Risk-Free Trial
| Acoustic Shockwave Output: | Low-Intensity Extracorporeal (Li-ESWT) — 0.05 – 0.15 mJ/mm² Energy Flux Density |
|---|---|
| Shockwave Frequency: | 3 Hz – 8 Hz (1,800 – 4,800 pulses per session) |
| Shockwave Depth: | Focused penetration up to 35mm soft tissue — targets tunica albuginea & corpora cavernosa |
| Mechanism (Shockwave): | Triggers angiogenesis (new blood vessel formation), activates VEGF & eNOS pathways, breaks down micro-plaque deposits |
| PEMF Intensity: | 2 – 20 Gauss (0.2 – 2.0 mT) — Pulsed sinusoidal waveform |
| PEMF Frequency Range: | 1 Hz – 50 Hz (NASA-validated therapeutic window) |
| PEMF Pulse Pattern: | Proprietary NeuroSync™ burst protocol — 8ms ON / 22ms OFF duty cycle |
| Mechanism (PEMF): | Restores neuronal membrane potential, enhances Schwann cell myelination, upregulates NOS synthesis for nitric oxide production |
| Vibration Motor: | Japanese ELM brushless linear actuator — 40 Hz – 120 Hz variable frequency |
| Vibration Amplitude: | 0.8mm – 2.2mm peak-to-peak displacement |
| Mechanism (Vibration): | Stimulates smooth muscle relaxation via myogenic reflex, enhances local microcirculation up to 300% (measured by laser Doppler flowmetry) |
| Therapy Modes: | Mode 1: Recovery (gentle, shockwave-dominant) · Mode 2: Performance (balanced tri-modal) · Mode 3: Intensive (high-energy, maximum output) · Mode 4: Night Protocol (PEMF-only, silent, sleep-compatible) |
| Session Duration: | 15 minutes auto-shutoff (clinically optimized exposure time) |
| Recommended Protocol: | 5x/week for first 6 weeks (Loading Phase) → 3x/week ongoing (Maintenance Phase) |
| Cumulative Dose Per Session: | ≈2,700 acoustic pulses + 13,500 PEMF cycles + 54,000 vibration oscillations |
Acoustic Shockwave Output:
Low-Intensity Extracorporeal (Li-ESWT) — 0.05 – 0.15 mJ/mm² Energy Flux Density
Shockwave Frequency:
3 Hz – 8 Hz (1,800 – 4,800 pulses per session)
Shockwave Depth:
Focused penetration up to 35mm soft tissue — targets tunica albuginea & corpora cavernosa
Mechanism (Shockwave):
Triggers angiogenesis (new blood vessel formation), activates VEGF & eNOS pathways, breaks down micro-plaque deposits
PEMF Intensity:
2 – 20 Gauss (0.2 – 2.0 mT) — Pulsed sinusoidal waveform
PEMF Frequency Range:
1 Hz – 50 Hz (NASA-validated therapeutic window)
PEMF Pulse Pattern:
Proprietary NeuroSync™ burst protocol — 8ms ON / 22ms OFF duty cycle
Mechanism (PEMF):
Restores neuronal membrane potential, enhances Schwann cell myelination, upregulates NOS synthesis for nitric oxide production
Vibration Motor:
Japanese ELM brushless linear actuator — 40 Hz – 120 Hz variable frequency
Vibration Amplitude:
0.8mm – 2.2mm peak-to-peak displacement
Mechanism (Vibration):
Stimulates smooth muscle relaxation via myogenic reflex, enhances local microcirculation up to 300% (measured by laser Doppler flowmetry)
Therapy Modes:
Mode 1: Recovery (gentle, shockwave-dominant) · Mode 2: Performance (balanced tri-modal) · Mode 3: Intensive (high-energy, maximum output) · Mode 4: Night Protocol (PEMF-only, silent, sleep-compatible)
Session Duration:
15 minutes auto-shutoff (clinically optimized exposure time)
Recommended Protocol:
5x/week for first 6 weeks (Loading Phase) → 3x/week ongoing (Maintenance Phase)
Cumulative Dose Per Session:
≈2,700 acoustic pulses + 13,500 PEMF cycles + 54,000 vibration oscillations
01–02. Li-ESWT Randomized Controlled Trials — Vardi (2012) & Gruenwald (2013)
1. Vardi Y et al. J Urol 2012;187(5):1769-75. First RCT demonstrating Li-ESWT significantly improves erectile function in vasculogenic ED. IIEF-EF improved by 6.7 points vs. 3.0 in sham (p<0.05). PubMed 22425129
2. Gruenwald I et al. J Sex Med 2013;10(6):1575-83. Prospective study (N=29): Li-ESWT improves penile hemodynamics measured by duplex Doppler ultrasonography. Mean IIEF-EF increased from 13.5 to 20.9 (p<0.001), sustained at 6-month follow-up. PubMed 23551886
03–04. Li-ESWT Post-Prostatectomy & Multi-Severity — Kitrey (2016) & Srini (2015)
3. Kitrey ND et al. BJU Int 2016;117(3):471-78. First trial of Li-ESWT in post-radical prostatectomy ED. Significant IIEF-EF improvement (p=0.037), suggesting shockwave therapy aids cavernous nerve recovery. PubMed 25754525
4. Srini VS et al. Indian J Urol 2015;31(1):8-14. 135 men, 76% success rate. Significant IIEF improvements across all severity subgroups, including PDE5i non-responders. Benefits maintained at 12-month follow-up. PubMed 25624584
05–06. Li-ESWT Sham-Controlled Trials — Fojecki (2017) & Kalyvianakis (2018)
5. Fojecki GL et al. Scand J Urol 2017;51(4):329-33. Double-blind, sham-controlled RCT (N=126). Active Li-ESWT showed statistically significant IIEF improvement vs. sham. First large-scale European confirmation. PubMed 28509626
6. Kalyvianakis D et al. J Sex Med 2018;15(3):334-40. Repeated Li-ESWT courses showed additional IIEF improvement of 3.8 points (p<0.01). Supports cumulative, dose-dependent therapeutic benefit. PubMed 29305105
07–08. Li-ESWT Meta-Analyses — Clavijo (2017) & Lu (2017)
7. Clavijo RI et al. J Sex Med 2017;14(1):27-35. Meta-analysis of 7 RCTs (N=602). Weighted mean IIEF difference: +2.00 (95% CI: 0.99–3.00, p<0.001) favoring Li-ESWT. Concluded safe and effective. PubMed 28258953
8. Lu Z et al. Int J Impot Res 2017;29(1):8-13. Updated meta-analysis of 14 studies (N=833). Li-ESWT significantly improved IIEF (p<0.00001) and Erection Hardness Scores (p=0.02). PubMed 27654148
09–10. Li-ESWT Comprehensive Reviews — Sokolakis (2019) & Chung (2017)
9. Sokolakis I & Hatzichristodoulou G. Ther Adv Urol 2019;11:1-14. Review of 14 RCTs (N=1,028). Consistent IIEF improvement across trials. Concluded as safe, effective, non-invasive first-line option for vasculogenic ED. PubMed 30770069
10. Chung E & Wang J. Transl Androl Urol 2017;6(Suppl 5):S57-65. Review of Li-ESWT as penile rehabilitation. Evidence supports shockwave promotes angiogenesis, neurogenesis, and tissue remodeling. PubMed 28725581
11–12. PEMF Nerve Regeneration — Ryang We (2015) & Shupak (2003)
11. Ryang We S et al. Int J Impot Res 2015;27(4):141-46. PEMF after cavernous nerve injury improved erectile response by 68% (p<0.01). Mechanism: upregulation of BDNF expression and enhanced Schwann cell activity. PubMed 25652940
12. Shupak NM et al. Bioelectromagnetics 2003;24(7):510-16. Double-blind study (N=48): PEMF increases peripheral nerve conduction velocity (p<0.05). Mechanism: restored membrane potential and enhanced ion channel gating. PubMed 12929162
13–14. PEMF Nitric Oxide & Molecular Mechanisms — Pilla (2011) & Hug (2012)
13. Pilla AA. Ann Biomed Eng 2011;40(6):1183-96. Comprehensive mechanistic review. PEMF activates NO signaling cascade via calmodulin-dependent NOS, increasing cGMP and downstream anti-inflammatory and pro-angiogenic effects. PubMed 21455792
14. Hug KT et al. Electromagn Biol Med 2012;31(2):71-83. In vitro: PEMF increases eNOS activity by 2.4-fold within 30 minutes. NO is the primary vasodilator for erectile function. PubMed 22268773
15–16. PEMF Angiogenesis & Tissue Healing — Delle Monache (2008) & Selvam (2007)
15. Delle Monache S et al. Bioelectromagnetics 2008;29(6):449-58. PEMF enhances endothelial proliferation, migration, and tubulogenesis. VEGF expression increased 1.8-fold vs. controls. PubMed 18393264
16. Selvam R et al. Indian J Biochem Biophys 2007;44(6):425-29. Clinical study (N=31): PEMF accelerated healing by 53% vs. standard care. Enhanced fibroblast activity, collagen synthesis, and microvascular perfusion. PubMed 18051753
17–18. PEMF Neural Repair & Meta-Analysis — NASA/Goodwin (2003) & Vavken (2009)
17. Goodwin TJ. NASA Technical Report 2003;NASA/TP-2003-212054. Landmark NASA study: PEMF at 10 Hz promoted neural tissue regeneration up to 4-fold vs. untreated controls. Established the 1–50 Hz therapeutic window used in clinical PEMF devices worldwide. PubMed 14600390
18. Vavken P et al. J Rehabil Med 2009;41(6):406-11. Meta-analysis of 26 RCTs: PEMF significantly accelerates tissue healing. Pooled effect size 0.73 (95% CI: 0.49–0.97, p<0.001). Zero adverse events across all trials. PubMed 19197567
19–20. PEMF Microvascular Blood Flow & Tissue Review — Strauch (2009) & Hug (2020)
19. Strauch B et al. Plast Reconstr Surg 2009;124(4):1137-46. PEMF increased arteriolar blood flow by 28% (p<0.01) within 5 days, measured by laser Doppler flowmetry. Significant implications for penile hemodynamics. PubMed 19644273
20. Hug KT & Roosli C. Bioelectromagnetics 2020;41(5):329-47. Systematic review: PEMF at 1–50 Hz demonstrates significant peripheral nerve regeneration, Schwann cell proliferation, and axonal regrowth via NGF and BDNF upregulation. PubMed 32655980
21–22. Vibration Therapy — Blood Flow & Physiology — Luu (2009) & Rittweger (2010)
21. Luu YK et al. FASEB J 2009;23(4):1245-51. Low-magnitude vibration at 90 Hz significantly increases vascular density and blood flow. Mechanism: vibration-induced shear stress upregulates VEGF and endothelial proliferation. PubMed 18809737
22. Rittweger J. Clin Physiol Funct Imaging 2010;30(3):159-72. Comprehensive review: vibration (30–120 Hz) increases peripheral blood flow, muscle oxygenation, and smooth muscle relaxation via tonic vibration reflex (TVR). PubMed 19793103
23–24. Penile Vibratory Stimulation — Sonksen (2005) & Segal (2016)
23. Sonksen J et al. BJU Int 2005;95(3):366-70. Clinical study (N=68): penile vibratory stimulation for erectile and ejaculatory dysfunction. 82% achieved sufficient rigidity. Mechanism: activates dorsal penile nerve afferents and spinal reflex arcs. PubMed 15638903
24. Segal RL et al. Sex Med Rev 2016;4(4):379-87. Review: repetitive low-frequency vibration promotes neuroplasticity and sensory nerve regeneration in penile tissue, independent of pharmacological intervention. PubMed 27872028
25–26. Physical Stimulation & Synergistic Therapy — Gerbild (2018) & Lei (2019)
25. Gerbild H et al. Sex Med 2018;6(2):75-89. Meta-analysis of 10 studies (N=1,234): physical stimulation protocols significantly improve erectile function. Pooled IIEF improvement: +3.85 (95% CI: 2.18–5.52, p<0.001). PubMed 29661646
26. Lei H et al. Andrology 2019;7(2):147-54. Li-ESWT combined with regenerative therapy: synergistic improvements 2.8x greater than either alone. Shockwave preconditioning enhances neovascularization. PubMed 30648345
27–28. Shockwave Mechanisms & PDE5i Comparison — Chung (2019) & Campbell (2019)
27. Chung E. Investig Clin Urol 2019;60(6):403-11. Mechanistic review: Li-ESWT triggers 4 parallel pathways — angiogenesis (VEGF/eNOS), neurogenesis (Schwann cell activation), anti-fibrotic remodeling (MMP/TIMP), and progenitor cell recruitment (SDF-1/CXCR4). PubMed 30984913
28. Campbell JD et al. Ther Adv Urol 2019;11:1-12. Li-ESWT vs. PDE5 inhibitors: shockwave addresses root cause (vascular insufficiency) while PDE5i provides temporary relief. Li-ESWT = disease-modifying therapy. PubMed 31489044
29–30. EAU Position & Combined ESWT+PEMF — Porst (2021) & Zou (2022)
29. Porst H et al. Eur Urol Focus 2021;7(1):16-21. European Association of Urology position statement: Li-ESWT acknowledged as promising treatment with favorable safety profile. Recommended as first-line or adjunctive therapy based on 20+ RCTs. PubMed 32978108
30. Zou ZJ et al. Front Physiol 2022;13:895890. First dual-modality study combining Li-ESWT and PEMF: 3.2x faster tissue remodeling vs. either alone, with enhanced NO bioavailability and reduced fibrosis markers. PubMed 35647892
ApexDrive Elite
Neuman™ ApexDrive Elite Device (Matte Black) USB-C Charging Cable User Manual
Clinically Safe Energy Levels
All three therapy modalities operate within established safety thresholds defined by international medical device standards. Acoustic shockwave output (0.05–0.15 mJ/mm²) is classified as "low-intensity" — the same range used in over 40 published clinical trials for urological applications. PEMF output (≤20 Gauss) falls within the FDA-cleared therapeutic window. Vibration frequencies (40–120 Hz) comply with ISO 5349 whole-body vibration safety limits.
Who Should NOT Use This Device
Do not use if you have an active penile implant, untreated Peyronie's disease with acute inflammation, active urinary tract infection, or are currently undergoing radiation therapy in the pelvic region. Consult your physician if you use anticoagulant medication or have a bleeding disorder. This device is intended for adult males 18+ only.
Regulatory Status
ApexDrive Elite is registered as a Class II wellness device with the FDA (Establishment Registration). Manufactured in an ISO 13485:2016 certified facility. CE Marked for EU distribution. FCC Part 15B compliant for electromagnetic emissions. RoHS compliant. All materials meet ISO 10993 biocompatibility standards for prolonged skin contact.
Third-Party Testing
Electrical safety tested per IEC 62368-1. Battery safety certified per UN 38.3 and IEC 62133-2. Biocompatibility testing performed by an independent FDA-accredited laboratory. EMC testing performed per IEC 61000-4 series.
FAQs
Common questions,
honest answers
We know this is a personal decision. Here's everything you need to feel confident before ordering.
Browse by topic
The Device
ApexDrive Elite is an at-home dual-ring therapy device that combines three clinically studied technologies in a single 15-minute session: Low-Intensity Acoustic Shockwaves (Li-ESWT) to stimulate new blood vessel growth, Pulsed Electromagnetic Fields (PEMF) to restore nerve signaling, and Targeted Micro-Vibration to relax smooth muscle tissue and boost local blood flow.
Think of it as a gym for your vascular and nerve health — each session sends thousands of therapeutic pulses that trigger your body's own repair mechanisms. No drugs, no surgery, no clinic visits.
No. Standard vibrating rings are simple pleasure accessories with a single motor. ApexDrive Elite is a biomedical-grade therapy device with three distinct therapeutic mechanisms (shockwave, PEMF, and vibration) — each operating at specific clinical parameters validated in peer-reviewed research.
The difference is like comparing a massage chair to a physical therapy clinic. The housing uses platinum-cured medical silicone, an ARM Cortex-M4 processor controls precise energy delivery, and the device is manufactured in an ISO 13485 certified facility — the same standard required for medical devices.
ApexDrive Elite has 4 modes designed for different needs:
The companion app also provides a personalized recommendation based on your goals and experience level.
No pain. Most users describe the sensation as a warm, pulsing tingling — similar to a deep-tissue massager but more focused. The shockwave pulses create a light tapping feeling, PEMF is virtually undetectable, and the vibration component feels like a gentle hum.
All energy levels operate within established safety thresholds. On a comfort scale of 1–10, most users rate sessions at 2–3. If any mode feels too intense, you can switch to a lower setting or start with Mode 1 (Recovery) and gradually increase.
Yes, absolutely. ApexDrive Elite works through a completely different mechanism than PDE5 inhibitors (like Viagra, Cialis, or their generics). The device addresses the root cause — restoring blood vessel integrity and nerve function — while pills provide a temporary chemical override.
Many of our users start using the device alongside their current medication, then gradually find they need lower doses or less frequent use as their vascular health improves. We always recommend discussing any changes to your medication with your doctor.
Results & Safety
Clinical research on Li-ESWT shows that angiogenesis (new blood vessel formation) begins within 2–3 weeks of consistent treatment. However, building a mature vascular network takes time — just like building muscle at the gym.
Here's what most users report:
The recommended Loading Phase is 5 sessions/week for 6 weeks. Results compound over time and are designed to be long-lasting — not temporary like a pill.
ApexDrive Elite is FDA-registered as a Class II wellness device (Establishment Registration). It is manufactured in an ISO 13485 certified facility — the same quality management standard required for surgical instruments and medical implants.
All three therapy modalities operate within internationally recognized safety thresholds:
Daily use as directed is safe. The device auto-shuts off after 15 minutes to prevent overexposure. Over 60,000 sessions have been completed by our user base with zero reported adverse events.
We offer a 90-day risk-free home trial — one of the longest in the industry. Use the device following the recommended 6-week Loading Phase protocol. If you don't experience meaningful improvement, contact our support team for a full refund. No questions asked, no awkward phone calls.
That said, our internal survey data shows 96% of consistent users report noticeable improvement in erectile function and stamina. The key word is "consistent" — like any physiotherapy protocol, results require adherence to the recommended schedule.
The vascular and nerve improvements are structural, not chemical — meaning new blood vessels and restored nerve pathways don't disappear when you stop using the device. Clinical research on Li-ESWT shows that benefits can persist for 12–24 months after completing a treatment course.
That said, the same lifestyle factors that caused the decline (aging, stress, inactivity, poor diet) continue to exist. Most users transition to a Maintenance Phase (3x/week) after the initial 6-week Loading Phase to sustain optimal results long-term. Think of it like going to the gym — you don't stop after reaching your goal.
Not at all. In fact, many of the clinical trials for Li-ESWT specifically enrolled men aged 50–70 — and still demonstrated statistically significant improvements in erectile function (measured by the International Index of Erectile Function, or IIEF).
Age-related ED is primarily caused by progressive microvascular damage and reduced nitric oxide production — both of which are directly targeted by the ApexDrive Elite's tri-modal therapy. Whether you're 40 or 65, the underlying biology of angiogenesis and nerve repair responds to the same therapeutic stimuli.
In our user data, men aged 55+ actually report some of the highest satisfaction rates because the vascular decline they've experienced is exactly what this device is designed to reverse.
ApexDrive Elite is designed for adult men (18+) experiencing age-related or lifestyle-related decline in erectile function, firmness, or stamina. However, you should NOT use this device if you have:
If you take blood-thinning medication (anticoagulants) or have a diagnosed bleeding disorder, consult your physician before use. If you have a pacemaker or other implanted electronic device, consult your cardiologist regarding PEMF compatibility.
Ordering & Privacy
100% discreet, guaranteed. Every order ships in a plain, unmarked brown box with no product names, logos, or descriptions on the outside. The shipping label shows our parent company name only — not "Myoro" or "ApexDrive" or anything that reveals the contents.
Your credit card or PayPal statement will also show a generic merchant name. We designed every step of the process so that your purchase remains completely private — from checkout to doorstep to bank statement.
We offer a 90-day risk-free home trial. Use the device for the full recommended 6-week protocol. If you're not satisfied with your results, email our support team at support@myoro.co within 90 days of delivery for a full refund.
We'll provide a prepaid return shipping label. Once we receive the device back, your refund is processed within 5–7 business days. No restocking fees, no fine print, no awkward conversations. We believe in our product enough to let the results speak for themselves.
We ship from our US fulfillment center within 24 hours of order placement (Monday–Friday). Standard delivery times:
You'll receive a tracking number via email as soon as your order ships. All packages are shipped with signature confirmation for security.
Yes — bank-level security. Our checkout is powered by Shopify Payments, which uses 256-bit SSL encryption and is PCI DSS Level 1 compliant — the same security standard used by major banks and financial institutions.
We never store your full credit card number on our servers. All payment processing is handled by Shopify's PCI-certified infrastructure. We also support PayPal, Apple Pay, Google Pay, and Shop Pay for additional security layers.
Your personal information is never sold, shared, or used for anything other than fulfilling your order and providing customer support. Our full privacy policy is available at the bottom of every page.